|
Describe and demonstrate some of the similarities andÄifferences between principles and values.5) Bill Alther is a zoologist who studies Anna's hummingbird.adults say they are more likely to make purchases If a codon is changes from ACC to CCC what would be the possible.what are the pros and con of patient privacy and security with the.It carries aĬurrent density of 7.65x10^6A/m^2, which. A copper wire has a diameter of 1.29x10^-3m.Creating an Economic Problem by yourself,and answer these four.expose the most important reasons why the location of small,.Please help me!! I'm stuck! And please show all the work.Space between gas molecules, which affects. chpt.9 pg 11: Pressure and temperature affect the amount of.Question 7 Arrange the following acids from weakest to strongest acid: H20 Butane 1-Butyne 1-Butene Water < Butane < 1-Butyne < 1-Butene 1-Butyne < 1-Butene < water Butane < 1-Butene < 1-Butyne < water Question 7 Arrange the following acids from weakest to strongest acid: H20 Butane 1-Butyne 1-Butene Water. What experimental observation allows you to arrange the amide and carboxylic acid functional.CH3CH2COOHPart C Arrange the following amines in order of decreasing base strength.1. HClOPart B Arrange the following carboxylic acids in order of decreasing acid strength.1. To rank items as equivalent, overlap them.1. Part A Arrange the following oxoacids in order of decreasing acid strength.Rank from strongest to weakest acid. Arrange the following oxoacids in order of decreasing acid strength.CF, CH, CH,COOH CF,CH,COOH CF,CH,CH,CH,COOH a. (3) Place the following carboxylic acids in the order from strongest acid to weakest acid. Place the following carboxylic acids in the order from Place the following carboxylic acids in the order from.To rank items as equivalent overlap them. Part C Arrange the following amines in order of decreasing base strength Rank from strongest to weakest base. Text solutions ( 1) Oxalic Acid is H OOC COOH, Malonic Acid is H OOC C H 2 COOH and Succinic Acid is H OOC (C H 2)2 COOH. To rank items as equivalent, overlap them. Part B Arrange the following carborylic acids in order of decreasing acid strength. Part A Arrange the following oxoacids in order of decreasing acid strength Rank from strongest to weakest acid. 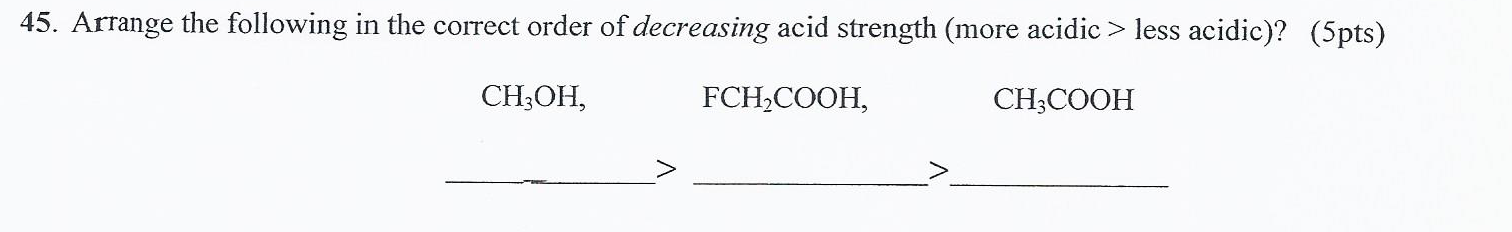 Hekp with steps please! Part A Arrange the following oxoacids in order of decreasing acid strength.Part C Arrange the following amines in order of decreasing base Part B Arrange the following carboxylic acids in order of Part A Arrange the following oxoacids in order of decreasing acid Part A Arrange the following oxoacids in order of decreasing acid strength.C0OH COOOH COOH COOH a b C The strongest acid is The second strongest is The third strongest is The weakest acid is Submit Answer Retry Entire Group 7 more group attempts remaining Indicate, by letter, the strongest acid and the weakest acid of those below. also a littleĪrrange the following carboxylic acids in order of acidity. Of them please would be much apreciated thanks.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
